EndoCem MTA
300mg/1package, Mixing ratio(P/L) = 300mg/0.12cc
ENDOCEM MTA is a revolutionary next generation MTA product based on the science of pozzolan reactions. featuring quick setting times, easy handling with consistent outcomes, excellent sealing capabilities and outstanding biocompatibility. ENDOCEM MTA exhibits high efficacy not only in apical retro-filling and root perforations but also in vital pulp therapies such as direct pulp capping.
Fast setting time · Adequate consistency
ENDOCEM MTA is non-miscible with liquid components, including blood, and thus is ideal for use in clinical situations where bleeding is difficult to control.
It has a fast setting time that allows for resin bonding to occur immediately after setting.

When used while pulpal bleeding occurs, a thin layer is applied first, and bleeding is controlled with cotton pellet. The remaining ENDOCEM MTA may then be applied. Setting may be confirmed by spraying water vigorously with an air/water syringe. Incomplete setting is identified when water spray washes away part of the material placed. If this is found to be the case the restoration should be removed in its entirety with the process started anew.
Mechanical property
ENDOCEM MTA maintains its integrity in less than ideal clinical situations, including contamination with blood or saliva and presence of inflammation or infection.
It is compatible with any type of bonding agents.
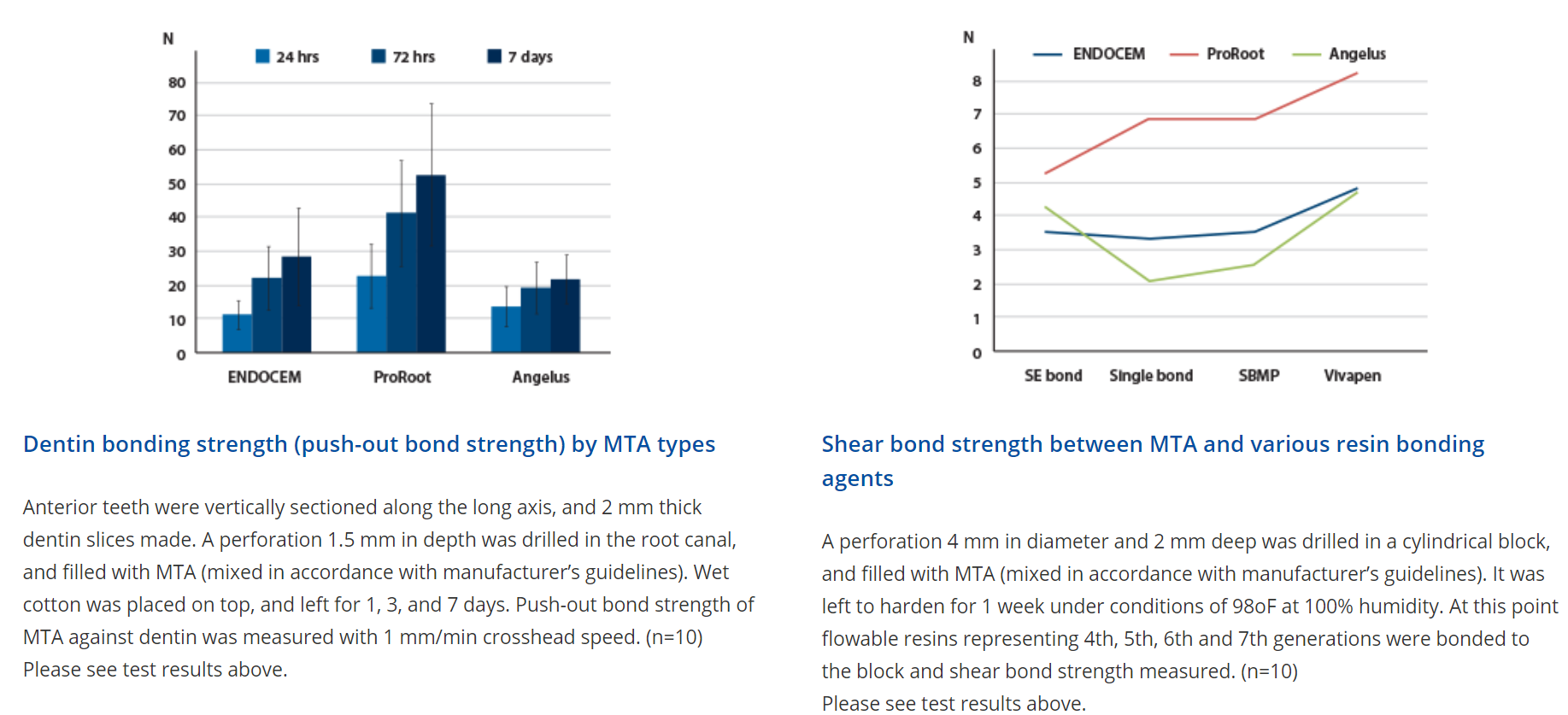
ENDOCEM MTA is seen as reaching a lower level of strength than ProRoot MTA initially. However, the bond strength increases over time.
Unlike conventional MTAs that exhibit bonding strength changes dependent on external factors such as saliva, blood, or inflammation, ENDOCEM MTA exhibits a consistent level of bond strength regardless of external factors.
ENDOCEM MTA exhibits consistent shear bond strength across all bonding agent types. This is further proof that ENDOCEM MTA is compatible with all bonding agents, and not adversely affected by specific characteristics of certain bonding agents.
Super sealing property
This is an example of stable clinical outcomes in presence of severe inflammation with minimization of dead space resulting from pozzolan reaction.

When (b) Proroot MTA is used for the same protocol the FBS infiltrated part was found to have been washed away during the setting process. When (c) IRM is used for the same protocol resistance to wash-out is comparable to that of (a) ENDOCEM MTA. However, many adjacent gaps are observed, and this is indicative of weaknesses where liquids, including water or blood, could possibly invade.
When exposed to liquids, i.e. saliva or GCF, the seal may be compromised over time due to degradation. Therefore, adding a resin base will be necessary before final restoration in direct pulp capping.
Least discoloration
ENDOCEM MTA allows teeth to maintain natural esthetics as it does not cause discoloration.
Picture view after tooth was filled with MTA and restored in artificial saliva.
Unlike (b) Proroot MTA or (c) Angelus MTA, (a) ENDOCEM MTA does not cause discoloration of adjacent tooth.
This is possible through the pozzolan reaction’s blockage of dentinal tubules. However, it should be noted that if leakage occurs in the final restoration, ENDOCEM MTA itself could undergo corrosion and that process may lead to discoloration.
Antibacterial Effect
ENDOCEM MTA has antimicrobial effects, not only on S. Mutans but also on E. Faecalis.

Enterococcus Faecalis is a pathogenic bacterium found with highest frequency in roots of endodontic failures, and is resistant against most MTA.
ENDOCEM MTA possesses strong anti-bacterial properties against E.Faecalis.
Excellent bio-compatibility
Our patented manufacturing techniques guarantee ENDOCEM MTA’s excellent biocompatibility.
SEM observation (x1000) of MG63 cells incubated for 72 hrs on (a)ENDOCEM MTA, (b)ProRoot MTA, (c)IRM
